Chemistry µGCSE:Alloys
10 quick questions - for GCSE and iGCSE
10 minutes maximum! Can you do it in 5? |
||
1. Mixtures of metals are called ...
| ||
2. Which diagram represents a solid alloy? |
||
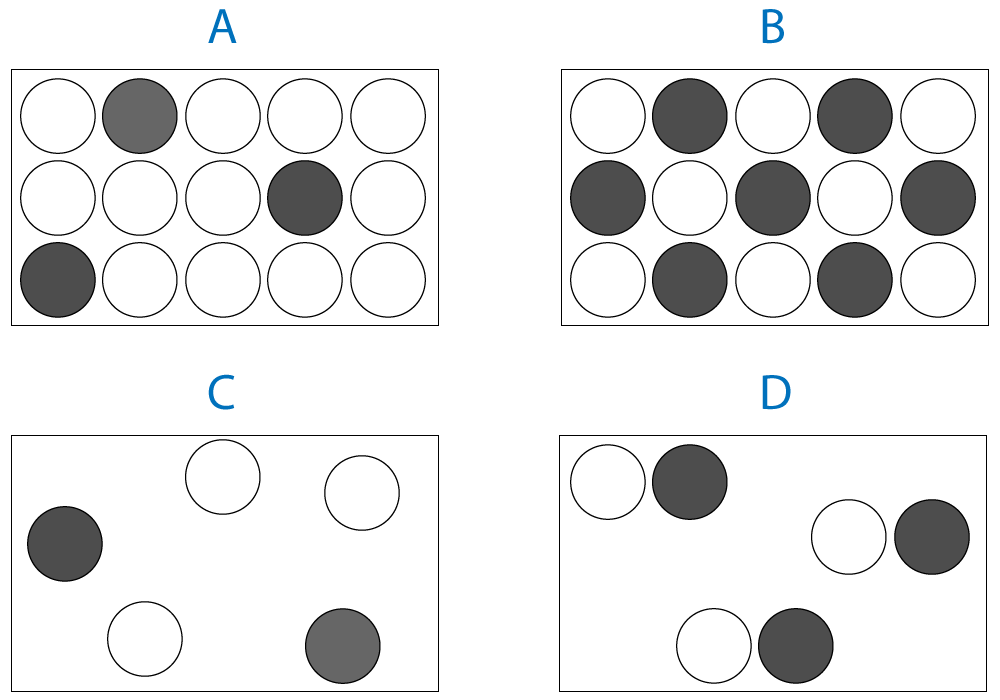 | ||
3. Mild steel is an alloy of iron and carbon. How does the carbon affect the properties of mild steel?
| ||
4. Iron and aluminium have many uses related to their properties. The metal most suitable to be used for the frame of a racing bicycle would be ... |
 |
|
| ||
5. Copper is used to make electrical wires but aluminium is preferred for overhead cables. The main reason that aluminium is used for overhead cables rather than copper is because aluminium ...
| ||
6. Brass is an alloy of copper and zinc. Which of the following statements is correct?
| ||
7. Copper is a transition element and is used to make saucepans. Which of the following properties of copper is NOT correct? |
 Mekonnen Wolday | CC BY-SA 4.0 |
|
| ||
8. The bodies of aircraft are often made from aluminium. |
 |
|
Which two properties of aluminum make it suitable for this use?
| ||
| 9. Stainless steel is an alloy of iron and other metals. It is strong and does not rust but it costs much more than normal steel. Which of the following would NOT be made from stainless steel?
| ||
10. The use of a metal is related to its properties. Which row below lists a property that is NOT correct for the use given?
| ||