Chemistry Edexcel GCSE:Earth+Atmosphere
Page 1
|
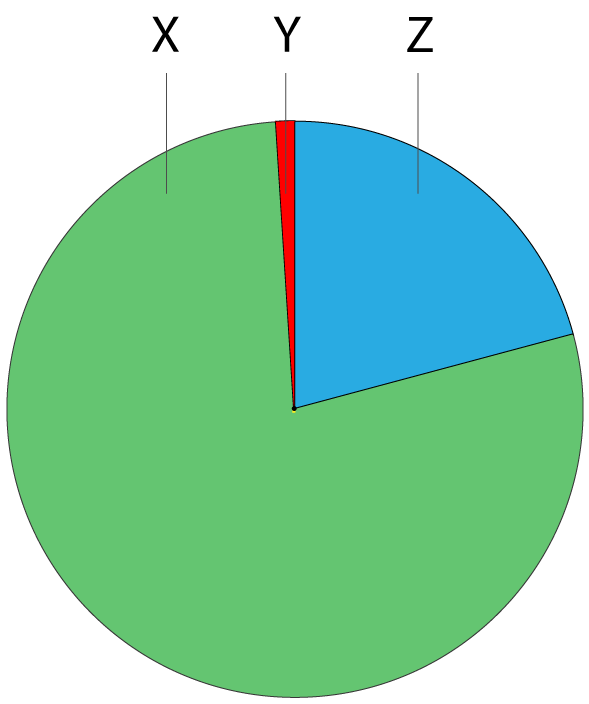
1. The pie chart shows the approximate percentage of the three most abundant gases in dry air in today’s atmosphere.
The gases represented by X, Y and Z are: |
 |
|||||||||||||||||||||
|
||||||||||||||||||||||
2. The test for oxygen gas is …
|
||||||||||||||||||||||
Q3-5: He obtained the following results:
|
|
|||||||||||||||||||||
3. The percentage of oxygen, to the nearest whole number, in the polluted air is..
|
||||||||||||||||||||||
| 4. What would be the effect, if any, on the calculated percentage of oxygen in the air if the student left the experiment set up for longer? | ||||||||||||||||||||||
| 5. What would be the effect, if any, on the calculated percentage of oxygen in the air if the student did not use excess iron wool? | ||||||||||||||||||||||
6. What process produced the gases in the Earth’s early atmosphere?
|
||||||||||||||||||||||
7. The composition of the Earth’s early atmosphere is uncertain but has been determined by analyzing evidence. Which of the following was the Earth’s early atmosphere NOT thought to contain?
|
||||||||||||||||||||||
| Q8-9: A sample of clean, dry air is passed over hot copper until all the oxygen in the air has reacted with the copper forming copper(II)oxide. |
||||||||||||||||||||||
|
||||||||||||||||||||||
8. What is the correct equation for the reaction occurring?
|
||||||||||||||||||||||
9. If the volume of air decreased by 20 cm3 what was the starting volume of the air?
|
||||||||||||||||||||||
10. Which of the following statements about the changes in the early atmosphere is NOT true?
|
||||||||||||||||||||||