NEED HELP? |
  |
|
10 minutes maximum! Can you do it in 5? |
||||||||||||
Q1+2. The diagram shows gas molecules trapped in a container. The molecules (particles) are moving very fast. they collide with the walls of the container producing a pressure. |
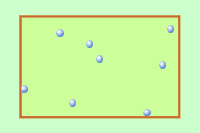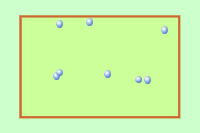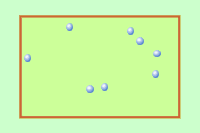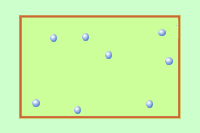 |
|||||||||||
1. Which of these statements best describes the effect of this pressure on the container?
|
||||||||||||
2. If the container volume is increased, without changing the temperature of the gas, what happens to pressure of the gas?
|
||||||||||||
3-6. A simple experiment is carried out using a gas syringe: A pressure sensor is attatched to the end of the syringe, and the pressure reading taken as the gas volume is reduced. The reading is taken after a while to allow the temperature to remain constant. The results are shown in the green table below. The formula linking pressure and volume is: p V = constant |
Database Center for Life Science (DBCLS) |
|||||||||||
3. What are the likely missing volume units labelled X in the results table below?
|
||||||||||||
4-6. What are the missing values in the results table for the syringe shown here?
|
||||||||||||
7-10. The volume of an air bubble in water is 60cm3 at a pressure of 4 x 105 Pa. The bubble expands as it rises to the surface. The formula linking pressure and volume is: p V = constant |
|
|||||||||||
7. Which of the following statements best describes the observations above?
|
||||||||||||
8. What will be the volume of the bubble in cm3 at a pressure of 1 x 105 pascals?
|
||||||||||||
9. During the bubble's ascent, what is the pressure of the water when the bubble has a volume of 80cm3?
|
10. Which of these graphs best shows the relationship between pressure and volume of a fixed mass of gas?
|
|||||||||||